Healy Device: A Class 2a Medical Device in Europe and Its Global Journey
The Genesis of Healy: Conceived as a Medical Device
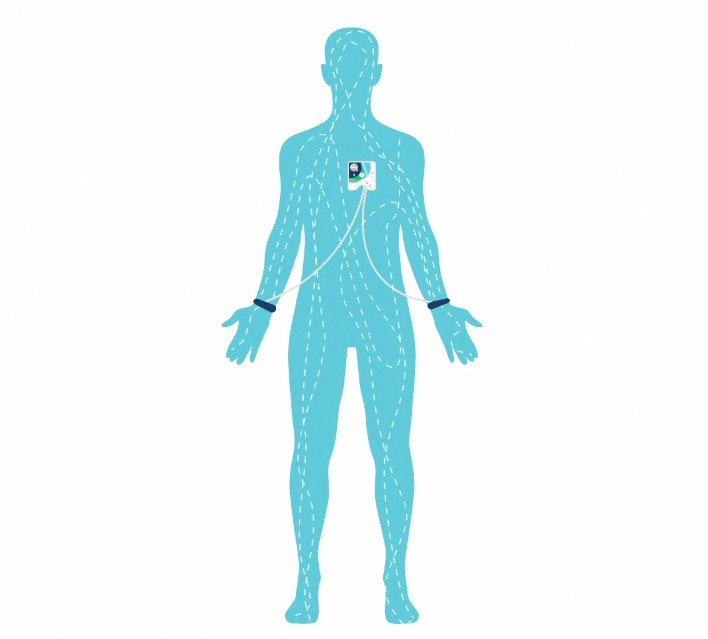
From its inception, the Healy Device was destined to be more than just another gadget; it was engineered to meet rigorous medical standards. Developed by German engineers specializing in medical devices, the journey began with meticulous planning and design. The entire development process was overseen by companies certified in medical device development, manufacturing, and assembly—all based in Germany.
This video describes in detail how the Healy Device became a class 2a medical device in Europe and its approval process as a wellness device in other countries around the world.
Certification: The Rigorous Path to Approval
Achieving Class 2a medical device status in Europe is no small feat. It involves a series of stringent tests and audits, conducted annually by notified bodies, to ensure compliance with the highest standards. For Healy, this included extensive electrical and electromagnetic safety tests, aligning with international standards for medical device safety.
Healy’s Clinical Validation
Before receiving approval, Healy was subjected to a comprehensive clinical evaluation as per the latest European guidelines. This evaluation included nearly 60 studies, demonstrating the device's efficacy in treating conditions like chronic pain, fibromyalgia, skeletal pain, migraine, and supporting mental health treatments for conditions such as depression and anxiety. The evaluation not only verified its effectiveness but also ensured its safety for public use.
Global Approvals and Varied Applications
Following its success in Europe, Healy gained approval in countries across the globe including the USA, Canada, New Zealand, and several others. Each country's approval process scrutinized Healy’s technical documentation and clinical evaluations afresh, adapting to local regulations which influenced the indications for which it could be approved.
A Wellness Device Around the World
In countries where Healy is not approved as a medical device, it takes on a different role as a wellness device. Known as "Healy Wellness," it offers programs to promote well-being, allowing users to benefit from the technology even without medical certifications.
Ongoing Research and Development
Healy World continues to advance its research, conducting post-market clinical follow-up studies to ensure continued efficacy and safety. These studies are crucial for maintaining certification and for evolving product efficacy. Although not all studies meet the stringent requirements for medical device approval, they still provide significant evidence of Healy's potential benefits in enhancing vitality and overall well-being.
The Future of Healy: Continuous Innovation and Global Expansion
The journey of the Healy Device is far from over. With ongoing studies and a commitment to high standards, Healy World aims to ensure that each of its products contributes positively to the physical, mental, and emotional wellness of its users. As Healy continues to evolve, it aims to not only meet existing standards but also to redefine what is possible in the convergence of health technology and holistic wellness approaches.
Healy: A Model of Medical and Wellness Innovation
The story of the Healy Device serves as a compelling example of how rigorous scientific processes, combined with innovative technology, can lead to significant advancements in both medical and wellness fields. As Healy continues to be recognized globally, it stands as a testament to the potential for technology to transform lives, demonstrating a successful balance between strict regulatory compliance and the pursuit of holistic health and wellness.
Huge savings on Healy Devices in Canada this April! Don't miss our Perfect Pair & Resonance bundles with free gifts. Limited time offer!
Healy World is rapidly expanding globally! Check our updated list of open markets in the Americas, Europe, and Asia Pacific to see if you can purchase a Healy device in your country. Find your official local shop now.
Exclusive Healy India sale! Don't miss incredible discounts on Healy Professional, Resonance, and valuable free accessories. Visit Healy Shop India!
Healy World India kicks off 2025 with amazing discounts! Grab a FREE MagHealy Professional with the Obsidian, save 40% on bundles, and get 30% off select Healy Editions. Plus, free coils and more! Limited-time offers end January 31st.
Discover Healy World's innovative supplement strips, Elements, as featured in L'Officiel England. Learn how these strips work with Healy's frequency technology to optimize your well-being.
Healy Colombia: New Year, New You! This January, experience the power of Healy frequency devices with incredible discounts! Save up to 45% on select editions and receive exclusive free gifts. Shop now and start your journey to a healthier, happier you!
Discover the transformative power of Solfeggio frequencies! Learn how the Solfeggio Frequency Music Player retunes your favorite songs into healing frequencies like 528 Hz and 432 Hz for relaxation, better sleep, and wellness.
For our UK customers, we’re offering an incredible 60% off January stock sale. Enjoy a 60% discount on a Healy Resonance Edition or MagHealy Professional Edition. With limited stock available, this exclusive offer won’t last long—contact us today to secure yours!
Don't miss out! Incredible Healy India promotions end soon! Grab a FREE MagHealy Pro or Healy Resonance Plus with select purchases. Plus, get up to 40% OFF Healy devices and bundles. Shop now before these deals disappear!
Celebrate the end of year with amazing Healy World promotions in Australia! Grab a Healy Device for yourself or a loved one with generous discounts and free gifts. Healy Obsidian Edition, Resonance, and more included in our Healy Sale!
The Healy Business Suite Module is your ultimate tool for personal growth and business success. With cutting-edge Information Field technology and four specialized modules, it empowers you to overcome challenges, optimize goals, and thrive at every stage of your career.
Exciting news! Healy, the revolutionary wearable wellness device, is now officially available in Cambodia. As of December 10th, 2024, Healy World Cambodia is open for business!
Get your Healy Device in the UAE! Shop via the Healy Europe store with direct shipping and access exclusive Healy discounts. Find prices for all editions, including Healy Obsidian, Healy Resonance, and Gold, along with the MagHealy Device.
Black Friday is here, and Healy India is celebrating with incredible deals! Get a FREE MagHealy Pro with Healy Obsidian, 50% off MagHealy Professional Bundles, 30% off Healy Editions, and more. Shop now!
Experience incredible Black Friday savings on Healy frequency technology in Singapore! Enjoy massive discounts on Healy devices and MagHealy bundles. Plus, receive exclusive free gifts with your purchase. Shop now and experience the Healy difference!
Don't miss Healy Colombia's Black Friday Sale! Save up to 60% on Healy Professional, MagHealy, and more. Limited-time offers on bioresonance devices for holistic wellness. Shop now!
Take advantage of Healy Canada’s Black Friday 2024 event with amazing deals all November! Save up to 50% on top devices like the Healy Obsidian, Resonance Plus, and Professional, plus receive exclusive gifts and financing options. Don’t miss these limited-time offers on Healy’s wellness technology
Discover our Healy Black Friday Sale in Mexico! Enjoy up to 55% off Healy devices, exclusive bundles, and free gifts at Healy Shop Mexico. Limited-time offers through November!
Take advantage of our exclusive Healy Obsidian Black Friday deal in the USA & Canada! Buy the Healy Obsidian Edition this November and receive a free MagHealy Professional, Meridian Module, and Black Yoga Jacket. This limited-time offer includes access to the VIP Obsidian Club and 0% financing for US buyers
Unlock 0% interest for up to 18 months on Healy Device purchases during our exclusive Black Friday promotion! Available only for US residents from November 1st to 11th, 2024, this limited-time offer allows you to bring home a Healy or MagHealy device with affordable monthly payments through Klarna. Don’t miss the best Healy prices in the USA—shop now on Healy Shop USA, select Klarna at checkout, and enjoy stress-free financing.
Grab incredible savings with Healy USA’s 2024 Black Friday Sale, running all November! From November 1st to 30th, seize discounts, free gifts, and exclusive bundles on top Healy devices like the Obsidian, Resonance Plus, and Professional. With up to 50% off, thousands in bonus gifts, and flexible financing options, now’s the perfect time to elevate your wellness journey. Shop early at the Healy USA Shop to secure the best Black Friday deals
Incredible limited-time offer! Buy a Healy Professional at 30% off and receive a FREE MagHealy Professional Edition. This amazing deal ends October 31st and is valid in Europe, the UK, and the UAE. Shop now through our Europe Shop!
Supercharge your frequency wellness journey with our incredible MagHealy Professional Bundle offer! Save over S$22,000 in our Healy Asia sale and experience the power of Healy Frequencies. Limited time only!
Enhance your Healy experience with our specially designed Healy Frequency Journal Prompts. These 45 prompts guide you in tracking your sessions, identifying the best frequencies, and reaching your wellness goals faster. Whether you're focused on improving sleep, boosting energy, or elevating your mood, these prompts help you reflect on your progress and optimize your results. Start today and unlock the full potential of your Healy device by journaling your journey toward better health and well-being!
Exclusive to customers in Singapore, Hong Kong, Taiwan, and Malaysia, save 60% on the MagHealy Professional with our limited-time promo code. Plus, receive a free Meridian Module! Don't miss this incredible opportunity to experience the power of PEMF therapy at an unbeatable price. Act fast – this offer ends soon!
¡Descubre las increíbles ofertas de Healy en Colombia este septiembre! Encuentra descuentos en dispositivos como Healy Obsidian, Healy Professional, Healy Resonance Plus y más. ¡No te pierdas la oportunidad de mejorar tu bienestar con Healy!
Ready to invest in your health and well-being? For a limited time, enjoy an exclusive 10% discount on the Healy Professional Edition with coupon code PROUSA. This offer is exclusively for USA residents. Don't miss out on this incredible opportunity!
Seize this limited-time opportunity! Get an EXTRA 10% OFF already discounted Healy devices with our exclusive Healy Canada coupon code. Healy Professional, Healy Resonance Plus, and MagHealy Professional are all included. Shop now and save! Offer ends Sept 30th, 2024.




























